Exhibition
by {0}
Full customization
The data is from past contract of the latest inspection report as assessed by independent third parties.
Annual export US $19,679,222
The data is from the latest inspection report assessed by independent third parties
Years in industry(23)
The data is from the latest inspection report assessed by independent third parties
Supplier assessment procedures
The data is from onsite check of the latest inspection report assessed by independent third parties
Full customization
The data is from past contract of the latest inspection report as assessed by independent third parties.
Annual export US $19,679,222
The data is from the latest inspection report assessed by independent third parties
Years in industry(23)
The data is from the latest inspection report assessed by independent third parties
Supplier assessment procedures
The data is from onsite check of the latest inspection report assessed by independent third parties
Full customization
The data is from past contract of the latest inspection report as assessed by independent third parties.
01
Best products
$799.00 /piece
2 pieces (Min.Order)
02
Best products
$8.80 - $11.00 /piece
10 pieces (Min.Order)
03
Best products
$999.00 - $1,500.00 /piece
2 pieces (Min.Order)
04
Best products
$750.00 - $950.00 /piece
2 pieces (Min.Order)
05
Best products
$109.89 - $141.90 /piece
20 pieces (Min.Order)





CATEGORY
Welcome to my shop
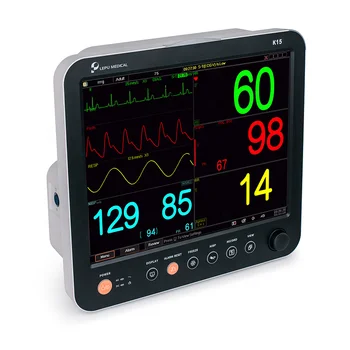
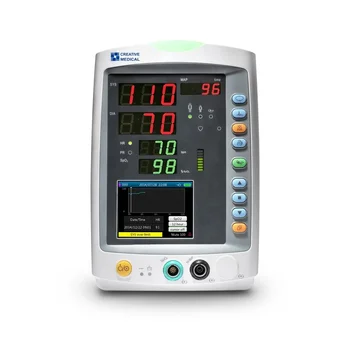
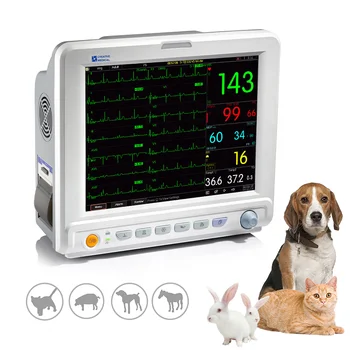
Patient Monitor
Oximeter
Electrocardiograph
Telehealth
US $600 - 700 /Piece
2 (Min. Order)
Audio Visual Alarms Factory Price High Quality Hospital ICU Vital Sign Monitoring Multiparameter Patient Cardiac MonitorUS $999 - 1,499 /Piece
US $500 - 800 /Piece
US $600 - 600 /Piece
US $1,599 - 1,600 /Piece
US $8.8 - 12.1 /Piece
10.0 (Min. Order)
Multifunctional automatic switch machine splash-proof and drop-proof low battery indicator removable pulse oximeterUS $109.89 - 141.9 /Piece
US $150 - 200 /Piece
US $9.79 - 10.89 /Piece
US $999 - 1,500 /Piece
2.0 (Min. Order)
Lepu Medical AI Smart Electrocardiogram Wireless Price EKG Monitor Device Leads 18 12 Channel Portable ECG Machine With TrolleyUS $650 - 750 /Piece
US $650 - 750 /Piece
1.0 (Min. Order)
Lepu Medical ECG-1112M The best-selling patient electrocardiographUS $500 - 600 /Piece
US $150 - 220 /Piece
10.0 (Min. Order)
Best Chinese Family Daily Measurement of Blood Pressure with Blue tooth Data Transmission Online Telemedicine DeviceUS $99 - 150 /Piece
US $199 - 399 /Piece

Lepu Medical AI Smart Electrocardiogram Wireless Price EKG Monitor Device Leads 18 12 Channel Portable ECG Machine With Trolley
$999.00 - $1,500.00
2 ( Min.Order )


2 ( Min.Order )
LEPU TH12 Medical Grade Telemedicine Ambulatory Preventice Cardiac 24 Hour Holter Heart Test ECG EKG Monitor$550.00 - $600.00


Why Choose Us
;Artificial intelligence "cardiogram analysis software"
The first domestic model to be approved for registration, currently the only one that has been approved by NMPA at the same time
FDA approved and CE certified artificial intelligence ECG products.
—— Passed the US FDA registration and approval
on November 19, 2018
—— Obtained the EU CE certificate on November 5, 2018
—— Obtained NMPA registration approval on January 22, 2020
AI-ECG Intellectual Property
—— 13 patents have passed the preliminary examination and 9 patents are under application;
—— 14 trademarks have passed the preliminary examination and 35 trademarks are in the process of application.
WHY CHOOSE US
Global Market